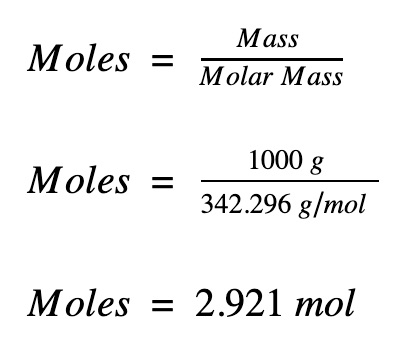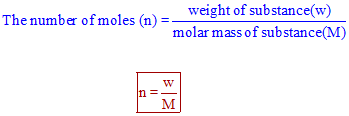How to Find Moles of Product from Moles of Reactant using a Chemical Equation | Chemistry | Study.com

Calculate the number of moles for the following: 52 g of He (finding mole from mass) 12.044 × 10^ 23 number of He atoms (finding mole from number of particles)

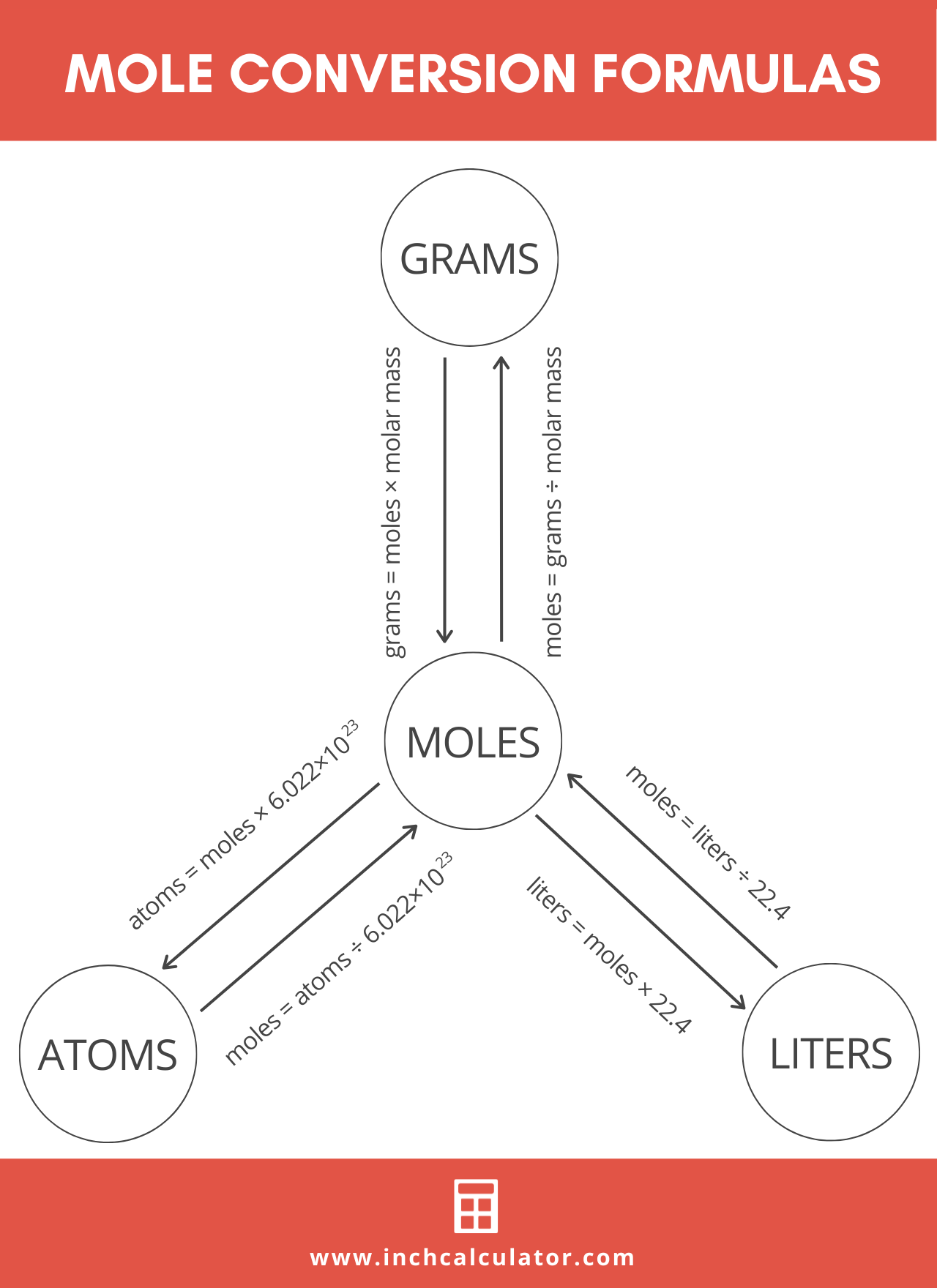
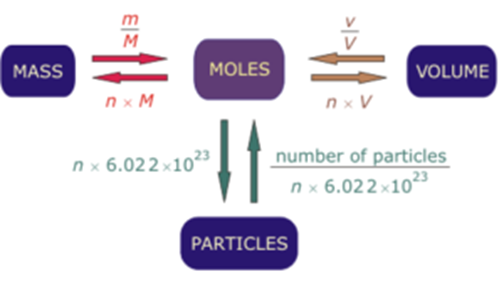
definition mole explained molar mass mol mols calculations how to read equations in moles deducing equations from reacting mole ratios questions gcse chemistry igcse KS4 science A level GCE AS A2 O
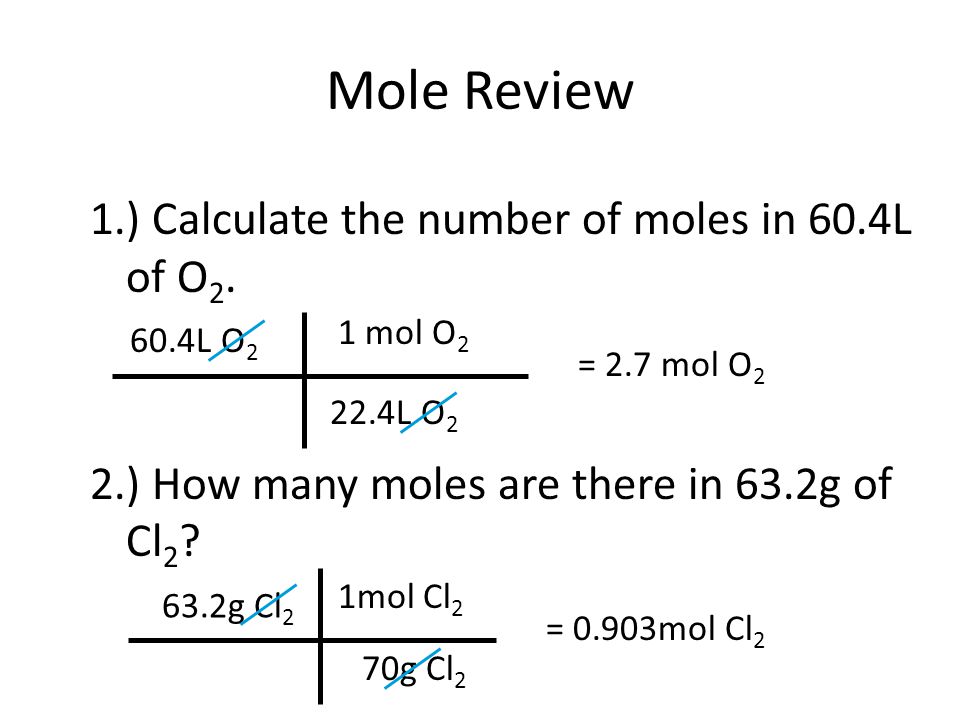
Mole Review 1.) Calculate the number of moles in 60.4L of O2. 2.) How many moles are there in 63.2g of Cl2? 1 mol O2 60.4L O2 = 2.7 mol O2 22.4L